“Our actions today will shape how people adapt and how nature responds to increasing climate risks.”
Hoesung Lee, IPCC Chairman
As mentioned last week, the United Nations’ Intergovernmental Panel on Climate Change (IPCC) published its most recent findings on the state of Earth’s climate. Over 900 scientists from 67 countries volunteered their time to contribute to the report, which focused on the measurable impacts of global warming. It contains 18 chapters about the different manifestations of climate change; six “cross-chapters,” which provide extra analysis; a Synthesis Report aka a general synopsis, and several “fact sheets” aka summaries of the chapters. My aim is to go through every sentence of this report that discusses the ocean and coastal areas (which is a pretty big portion of it).
Last week we learned about sea level rise, specifically why it is important to everyone and how it will worsen as our planet warms. After reading through a couple more IPCC chapters about risks to small islands and ocean & coastal ecosystems, a phenomenon that kept popping up was ocean acidification. The IPCC defines it as “a process [that] increases the concentrations of aqueous CO2, bicarbonate and hydrogen ions, and decreases pH, carbonate ion concentrations and calcium carbonate mineral saturation states...” Unless you speak science or have an advanced degree in chemical oceanography, after reading that definition you probably look like this:
So what is ocean acidification, and why are scientists concerned about it? Here are the ~basics~:
What is ocean acidification?
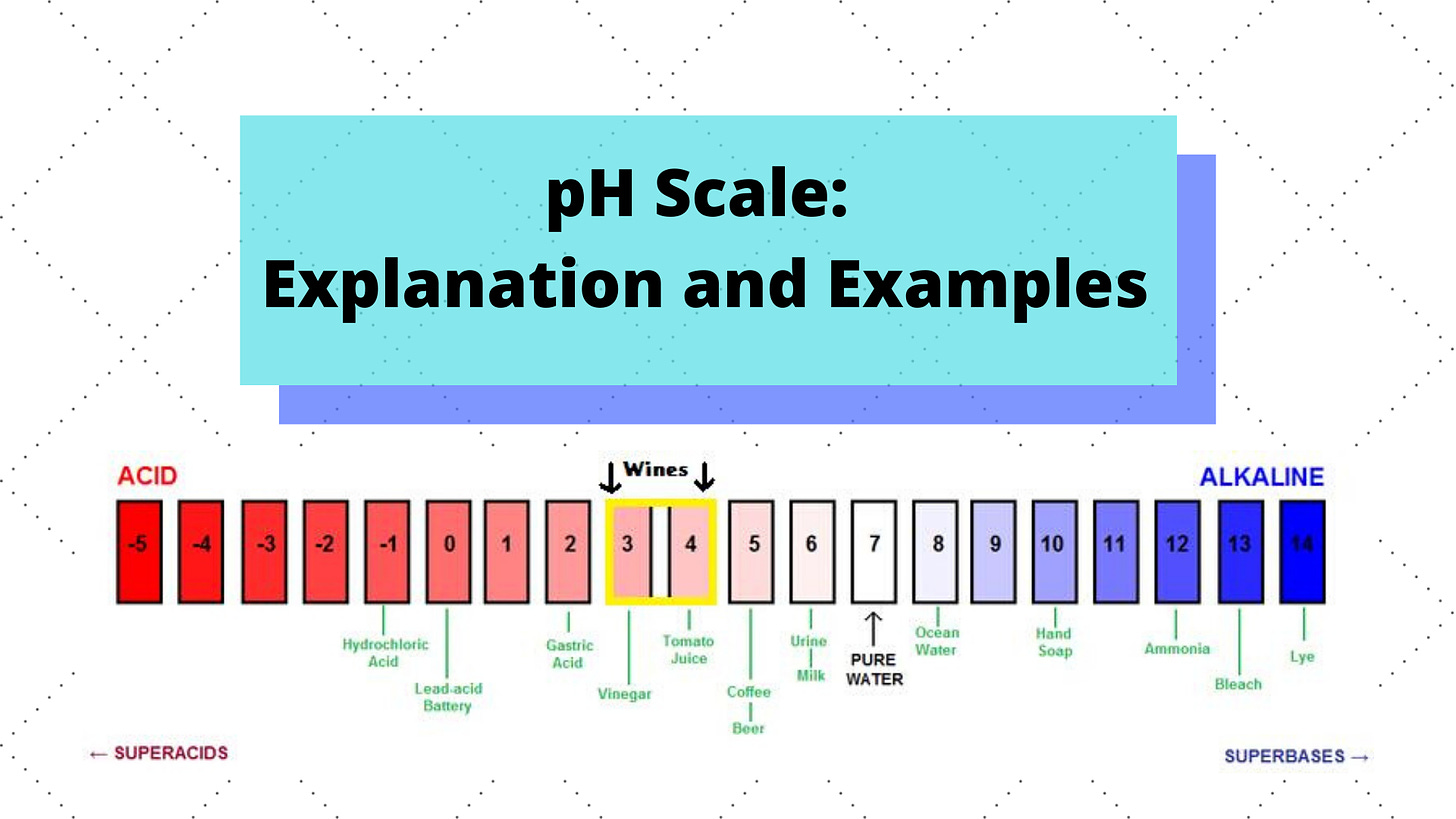
Ocean acidification (OA) is the process by which sea water increases in acidity. Acids and bases are measured on the pH scale, which runs from 0-14 with 7 being neutral like tap water. The “pH” stands for “potential of hydrogen” — Element #1 is found in most liquids and solutions (e.g. water = H2O) in the form of a positive ion aka a little active/reactive particle. The pH scale is just a measurement of how active these hydrogen ions (H+) are, and if there are more H+ ions the substance is more acidic.
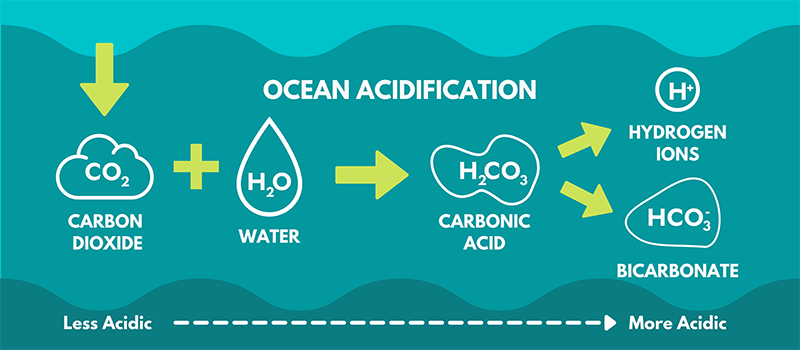
Seawater in the pre-industrial times had an average pH of 8.2 (nowadays it’s 8.1), which means it was slightly more basic and had fewer H+ ions than tap water. This is because it has high amounts of elements & ions freely floating around. One of the most important ones is calcium, which bonds with aqueous(dissolved in the water) carbon dioxide to form calcium carbonate, a key building block for shells and corals. Carbon dioxide (CO2) appears in the ocean by respiration, hydrothermal vents, and other natural processes. However, the oceans absorb 1/3rd of the carbon dioxide that we humans emit into the atmosphere. There is now more CO2 in the ocean than can bond with the calcium ions, and this extra CO2 makes seawater more acidic because it increases the number of H+ ions. Here’s how that works:
When CO2 is dissolved in seawater, it bonds with the freely-floating hydrogens (H2) and oxygen (O) molecules to create carbonic acid, symbolized as H2CO3.
This H2CO3 dissipates into little ions of its own, creating bicarbonate (HCO3) and a solitary H+ ion. Bicarbonate is useless to coral & shelled organisms — because it has already bonded with H, it cannot bond with Ca.
This HCO3 further dissipates into yet another H+ ion and then finally carbonate (CO3), which can finally bond with the freely-floating calcium ions to form calcium carbonate (CaCO3), an essential substance for shelled organisms.
So, seawater’s freely-floating, NEUTRAL H2 is split into two H+ ions by the addition of extra CO2. As you can see this hinders the process of forming CaCO3 and increases the acidity of the water.
Who and what is affected by OA?
Anytime there is a change in the ocean’s pH, calcification rates (how effectively animals can build their shells) are going to be affected. Calcite is the mineral form of calcium carbonate; it is used by plankton and invertebrates like echinoderms (sea stars & urchins) to form shells or skeletons. Marine life that depends on calcite/CaCO3 will suffer, as will fish, mammals, & other animals up the food chain that are reliant on shelled organisms. Those that do not get the calcite they need to survive will dissolve; as the severity of OA increases, there will be more widespread extinction events of marine species. Therefore, people who rely on shellfish(ing) as well as those who live near coral reefs or on small remote islands will notice the effects of OA much sooner than inland or high-elevated communities. As stated plainly in the IPCC report, if the climate warms beyond 1.5 degrees Celsius as it is predicted to do during this decade, acidification and higher seawater temperatures “would have a detrimental effect on the livelihoods of 200 million fishermen.” Since the fisheries industry makes up a huge portion of many small islands’ GDP, these extinction events will hinder economic growth, both locally and globally.
So, ocean acidification will disrupt all levels of the marine food chain and negatively impact the global economy. How will it worsen as the climate changes?
OA & climate change
Anthropogenic changes (aka those made by humans) have caused “unprecedented changes” in our oceans, exposing plants and animals to sub-optimal conditions. Indeed, OA is already affecting marine ecosystems. Ocean surface pH has declined over the past four decades, and it is predicted to continually decrease at a steady rate as the 21st century goes on. Previously the average pH decrease is 0.01/decade, but tropical regions will see their pH decrease at a much faster rate (0.016-0.02/decade). These areas of the ocean, home to coral reefs & an enormous amount of biodiversity, experience the largest exchange between the ocean and atmosphere because of the constant evaporation & condensation around the equator (think tropical rainforest, but on the ocean’s surface). Additionally, polar regions are expected to be hit the hardest by OA in future decades: the pH of the Arctic Ocean is predicted to decrease by as much as 0.026 in the coming years. This is because the colder, denser waters are very nutrient-rich, but as more CO2 is emitted & subsequently absorbed, the areas will become undersaturated with calcium carbonate. The IPCC states in the report that in 20 years the impacts of OA will be too large to ignore.
Interestingly, acidification is not directly affected by temperature changes. OA will continue to happen even if the planet suddenly stops heating up — why? While all greenhouse gasses warm the climate, carbon dioxide is the one that is directly causing OA. Therefore, Only a reduction in CO2 emissions would combat OA. The report specifically states that geoengineering techniques aiming to reduce solar radiation will have no effect on OA because they do not solve the problem of carbon dioxide (CO2) emissions. Atmospheric CO2 levels have increased by over 45% in the past two centuries, from roughly 280 parts per million to 418 ppm:
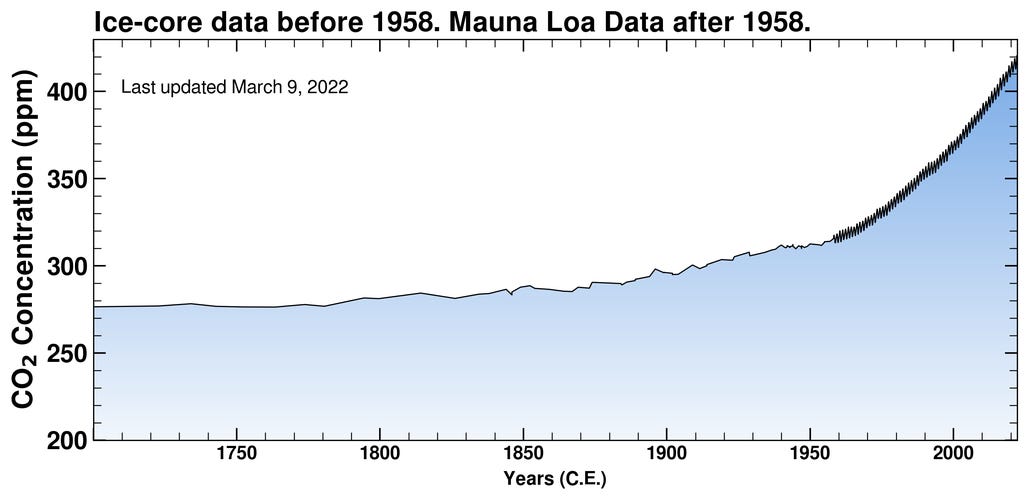
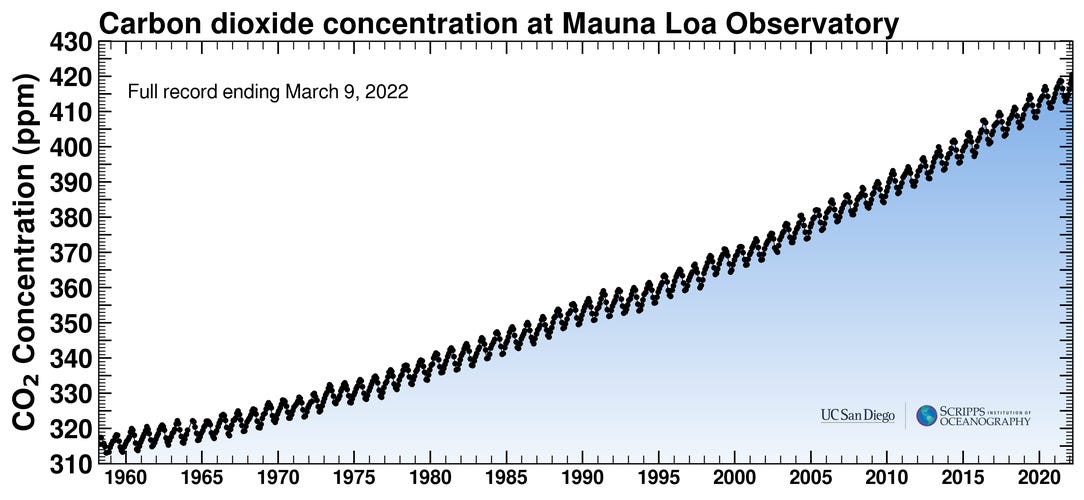
Yet scientists did not begin to study OA until 2003, when the rapid shift in seawater pH became noticeable and the term “ocean acidification” was coined. Towards the end of the Small Islands chapter, the IPCC mentions that there are still many unknowns about the long-term affects of OA. In a list of information that was lacking and needs to be studied more, the IPCC includes “impacts… on species, including relationship to disease outbreaks, and changing breeding grounds of marine species and impacts on fisheries & marine-based livelihoods.” Basically, no one has published anything, let alone anything conclusive, about the vulnerability of island ecosystems and marine environments to OA.
Where & when will these changes be noticed?
These changes in seawater pH are already manifesting themselves in tropical and polar ecosystems — far sooner than predicted by previous reports. Coastal waters are already experiencing a great amount of change due to runoff, acid rain, and regional variations; all those phenomena can mask the impacts of OA. But shallow surface waters are not the only areas affected by OA, however; with vertical mixing along the water column, OA is spreading deeper into the ocean and impacting the survival of even the smallest organisms. Instead of giving you a long list of animals that could/can/will be affected by acidification, I think the plight of one little species can illustrate the dangers facing all. Let us examine pteropods, tiny shelled zooplankton nicknamed ‘sea butterflies.’
Tricia Thibodeau, the professor who taught my college ocean acidification class, focused on the response of pteropods to ocean acidification. She took samples from Antarctica and compared the thickness of the pteropods’s shells with the overall acidity of the water. (I used to work in her lab and was in charge of sorting through the tiny marine biota & picking out these pteropods. I had to be SUPER gentle, for their shells were so delicate that a little too much pressure could break them & her study would be ruined!) As, after reading all about OA, you will be unsurprised to hear, Tricia discovered that pteropods are constantly rebuilding their protective shells to avoid dissolving. This repeatedly stressed the animals and leads to a premature death — that is, if they do not dissolve into the surrounding seawater first. Thus acidification is causing unique physical and mental harm to marine life; you can read more of her research here.
Can we stop this? If not, how can we adapt?
The only way to stop OA is to halt all greenhouse gas emissions. If we stop emitting greenhouse gases, the ocean would not absorb as much excess CO2. That could hinder or halt OA, but so far only two countries (Bhutan and Suriname) have gone carbon neutral, and only six more have pledged to relinquish their dependence on fossil fuels by mid-century:
Even then, we will still need to adapt to current conditions by preserving coral reefs and finding a way to deacidify the water (new geoengineering project anyone?). The current IPCC report offers little guidance on this, for there is no way to remove excess carbon from the ocean on a small-scale, regional basis. Unless global leaders take swift action to reduce CO2 emissions, we’ll have to be content with eating less fish & shellfish at the very least in the decades to come.
Stay tuned for next week, when I’ll discuss coral bleaching (unless something more interesting appears in the IPCC chapters) and how it’s affected by ocean acidification & temperature increases.
Sources not already linked in paragraphs or graphics above:
-Cooley, S., D. Schoeman, L. Bopp, P. Boyd, S. Donner, D.Y. Ghebrehiwet, S.-I. Ito, W. Kiessling, P. Martinetto, E. Ojea, M.-F. Racault, B. Rost, and M. Skern-Mauritzen, 2022: Ocean and Coastal Ecosystems and their Services. In: Climate Change 2022: Impacts, Adaptation, and Vulnerability.Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change [H.-O. Pörtner, D.C. Roberts, M. Tignor, E.S. Poloczanska, K. Mintenbeck, A. Alegría, M. Craig, S. Langsdorf, S. Löschke, V. Möller, A. Okem, B. Rama (eds.)]. Cambridge University Press. In Press.
-Mycoo, M., M. Wairiu, D. Campbell, V. Duvat, Y. Golbuu, S. Maharaj, J. Nalau, P. Nunn, J. Pinnegar, and O. Warrick, 2022: Small Islands. In: Climate Change 2022: Impacts, Adaptation, and Vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change [H.-O. Pörtner, D.C. Roberts, M. Tignor, E.S. Poloczanska, K. Mintenbeck, A. Alegría, M. Craig, S. Langsdorf, S. Löschke, V. Möller, A. Okem, B. Rama (eds.)]. Cambridge University Press. In Press.
-O'Neill, B., M. van Aalst, Z. Zaiton Ibrahim, L. Berrang Ford, S. Bhadwal, H. Buhaug, D. Diaz, K. Frieler, M. Garschagen, A. Magnan, G. Midgley, A. Mirzabaev, A. Thomas, and R. Warren, 2022: Key Risks Across Sectors and Regions. In: Climate Change 2022: Impacts, Adaptation, and Vulnerability.Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change [H.-O. Pörtner, D.C. Roberts, M. Tignor, E.S. Poloczanska, K. Mintenbeck, A. Alegría, M. Craig, S. Langsdorf, S. Löschke, V. Möller, A. Okem, B. Rama (eds.)]. Cambridge University Press. In Press.